Description
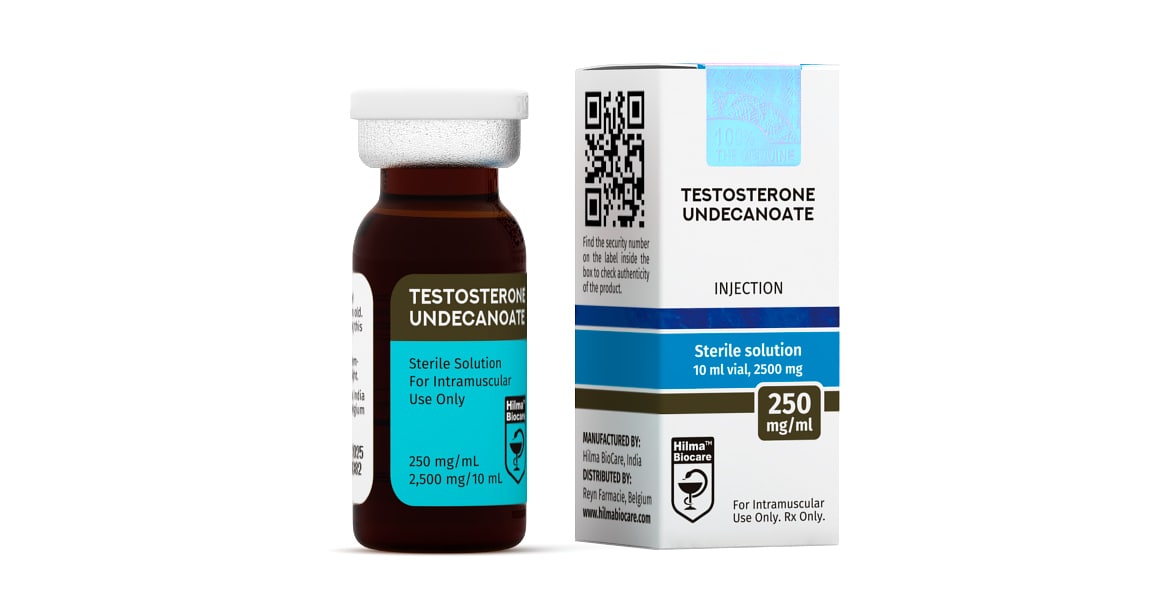
Testosterone Undecanoate
Strength: 250 mg/ml
Molecular Formula: C30H48O3,
Molecular Weight: 456,7003 g/mol
Active Ingredient: Testosterone Undecanoate
CAS number: 5949-44-0
Dosage Form: Injectable, oil base sterile solution
Route: Injection
Market Status: Prescription
Company: Hilma Biocare
DESCRIPTION
Testosterone undecanoate (17beta;-undecyloxy-4- androsten-3-one) is an unsaturated,
aliphatic, fatty acid ester of T. Testosterone undecanoate for intramuscular administration
provides a long-acting testosterone depot (half-life of 33.9 +/- 4.9 days) with favorable
pharmacokinetic properties. In hypogonadal men, 1000 mg of Testosterone undecanoate
can maintain stable plasma T levels for 12 weeks.
DIRECTIONS TO USE
Testosterone undecanoate is indicated for replacement therapy in adult males with
conditions that are linked with an absence or deficiency in endogenous testosterone
production. Testosterone undecanoate also helps to maintain muscle and prevent bone loss,
and is necessary for natural sexual ability/desire. Testosterone undecanoate may also be
used in certain adolescent boys to cause puberty in those with delayed puberty. This product
should not be used by women.
INDICATIONS
Primary hypogonadism (congenital or acquired): testicular failure due to cryptorchidism,
bilateral torsion, orchitis, vanishing testis syndrome, orchiectomy, Klinefelter’s syndrome,
chemotherapy, or toxic damage from alcohol or heavy metals. These men usually have low
serum testosterone concentrations and gonadotropins (follicle-stimulating hormone (FSH],
luteinizing hormone (LH)) above the normal range.
Hypogonadotropic hypogonadism (congenital or acquired): gonadotropin or luteinizing
hormone-releasing hormone (LHRH) deficiency or pituitary- hypothalamic injury from tumors,
trauma, or radiation. These men have low testosterone serum concentrations but have
gonadotropins in the normal or low range.
CONTRAINDICATIONS
Men with carcinoma of the breast or known or suspected carcinoma of the prostate.
SIDE EFFECTS
Because clinical trials are conducted under widely varying conditions, adverse reaction rates
observed in the clinical trials of a drug cannot be directly compared to rates in the clinical
trials of another drug and may not reflect the rates observed in clinical practice. Testosterone
Undecanoate was evaluated in an 84-week clinical study using a dose regimen of 750 mg (3
mL) at initiation, at 4 weeks, and every 10 weeks thereafter in hypogonadal men. The most
commonly reported adverse reactions (>2%) were: acne (5.2%), injection site pain (4.6%),
prostate specific antigen increased (4.6%), hypogonadism (2.6%) and estradiol increased
(2.6%). Adverse events attributable to pulmonary oil microembolism and anaphylaxis were
reported in a small number of patients in controlled clinical trials. The following adverse
reactions have been reported: premature closure of bony epiphyses with termination of
growth, and precocious puberty.
PRECAUTIONS
Before using Testosterone undecanoate, tell your doctor or pharmacist if you are allergic to
it; or if you have any other allergies. This product may contain inactive ingredients (such as
benzyl benzoate, refined oil), which can cause allergic reactions or other problems. Before
using this medication, tell your doctor or pharmacist your medical history, especially of:
cancer (especially prostate or breast cancer), prostate problems (such as enlarged prostate),
heart problems (such as heart failure, previous heart attack), stroke, kidney problems, liver
problems, high cholesterol, difficulty breathing during sleep (sleep apnea), blood clots. If you
have diabetes, this product may lower your blood sugar. Check your blood sugar regularly
as directed and share the results with your doctor. Tell your doctor right away if you have
symptoms of low blood sugar, such as sudden sweating, shaking, fast heartbeat, hunger,
blurred vision, dizziness, or tingling hands/feet. Your doctor may need to adjust your
diabetes medication, exercise program, or diet. Some men, particularly older adults, are at a
higher risk for developing enlarged prostate or prostate cancer while using this medication.
Your risk of prostate problems should be checked by your doctor before you start
testosterone.
DRUG INTERACTIONS
Insulin
Changes in insulin sensitivity or glycemic control may occur In patients treated with
androgens. In diabetic patients, the metabolic effects of androgens may decrease blood
glucose and, therefore, may necessitate a decrease in the dose of anti-diabetic medication.
Oral Anticoagulants
Changes in anticoagulant activity may be seen with androgens, therefore more frequent
monitoring of international normalized ratio (INR) and prothrombin time are recommended in
patients taking warfarin, especially at the initiation and termination of androgen therapy.
Corticosteroids
The concurrent use of testosterone with corticosteroids may result in increased fluid
retention and requires careful monitoring particularly in patients with cardiac, renal or hepatic
disease.
DOSAGE AND ADMINISTRATION
The recommended dose of Testosterone Undecanoate is 750 mg injected intramuscularly,
followed 750 mg injected after 4 weeks, then 750 mg injected every 10 weeks thereafter. To
help you remember, use a calendar to mark the days you will receive an injection.
Misuse or abuse of testosterone Can cause serious side effects such as heart disease
(including heart attack), stroke, liver disease, mental/mood problems, abnormal drug-seeking
behavior, or improper bone growth (in adolescents).
Do not increase your dose or use this drug more often or for longer than prescribed. When
testosterone is misused or abused, you may have withdrawal symptoms (such as
depression,irritability, tiredness) when you suddenly stop using the drug.
These symptoms may last from weeks to months.
PRESENTATION
Testosterone Undecanoate 2500 IC mg/ml, 10 ml multiple dose vial.
STORAGE
Store in a cool dry place between 15 – 25°C. Protect from light.