Description
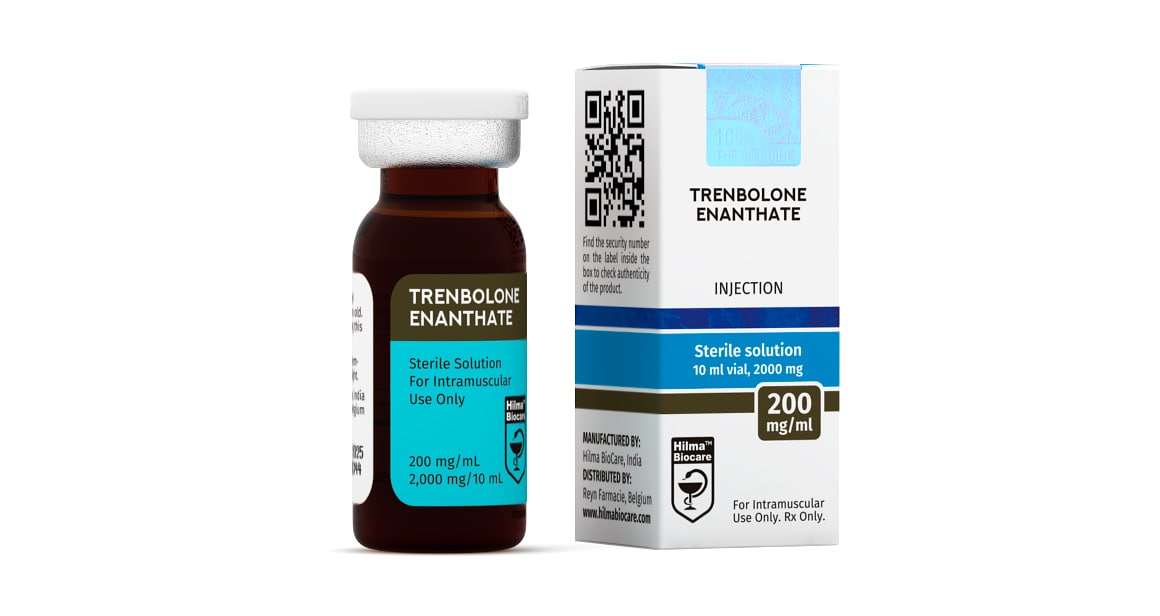
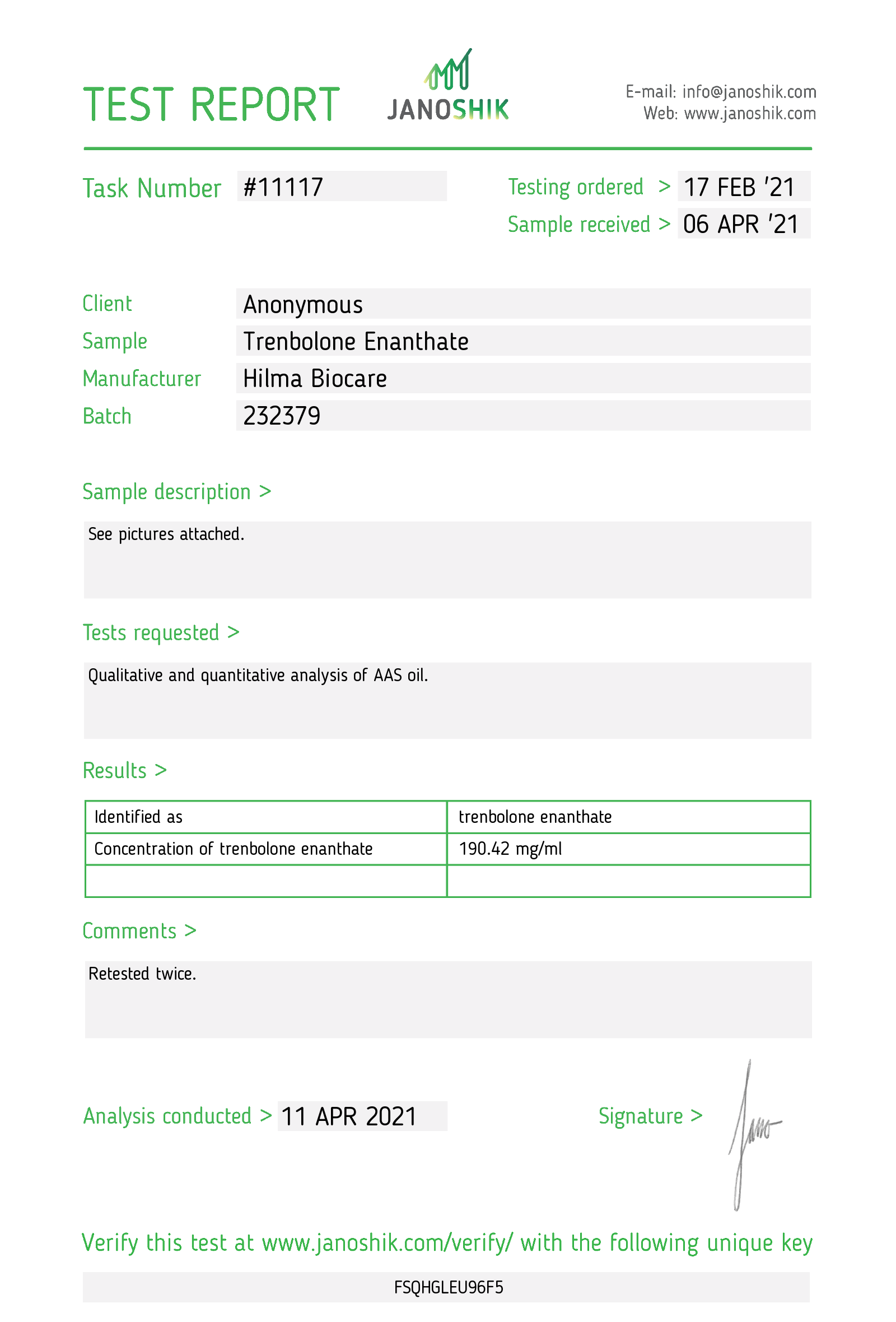
Trenbolone Enanthate
Strength: 200mg/ml
Molecular Formula: C18H2202.
Molecular Weight: 270.37 g/mol
Active Ingredient: Trenbolone enanthate
CAS number: 10161-33-8
Dosage Form: Injectable, oil base sterile solution
Route: Injection
Market Status: Prescription
Company: Hilma Biocare
DESCRIPTION
Trenbolone Enanthate 200 is an oil based solution of trenbolone enanthate for IM injection.
Trenbolone is an anabolic steroid with significant anabolic and androgenic effects. The
enanthate ester produces an initial serum response within 24-hours with a duration of action
of 7 to 10 days. Trenbolone promotes significant increases in strength, muscle anabolism,
appetite, and aggression and has been demonstrated to reduce body fat.
INDICATIONS
Males: Trenbolone use may be indicated in patients where substantial weight gain and
increases in musculature are required for patient health after substantial losses of body
mass especially in instances where caloric intake is limited and other anabolic therapies
have previously failed. The physician and patient must consider the risks of therapy versus
the potential benefits.
CLINICAL PHARMACOLOGY
Anabolic steroids are synthetic derivatives of testosterone. Certain clinical effects and
adverse reactions demonstrate the androgenic properties of these drugs. Complete
dissociation of anabolic and androgenic effects has not been achieved. The actions of
anabolic steroids are thus similar to those of male sex hormones. Anabolic steroids suppress
the gonadotropic functions of the pituitary and may exert a direct effect upon the testes.
During exogenous administration of anabolic androgens, endogenous testosterone release
is inhibited through inhibition of pituitary luteinizing hormone (LH). At large doses,
spermatogenesis may be suppressed through feedback inhibition of pituitary folliclestimulating hormone (FSH). Trenbolone binds strongly to the androgen receptor and its
action is generally considered to derive therefrom. It produces significant anabolism even
during periods of limited caloric restriction. The literature offers conflicting reports of the
susceptibility of trenbolone to aromatize into estrogen or reduce to a dihydrotestosterone
derivative. Rat studies have suggested that trenbolone may exert effects
dihydrotestosterone through a non- DHT pathway potentially with direct receptor action. It
has been suggested that trenbolone may reduce cortisol production through an
indeterminate pathway of activity upon glucocorticoid receptors.
Trenbolone has been demonstrated to promote muscle growth, appetite, aggression, and
the production of red blood cells through the production of erythropoietin stimulating factors.
Trenbolone is suspected of selective binding of the progesterone receptor potentially acting
as both an agonist and an antagonist. It has been suggested that trenbolone is capable of
binding to the prolactin receptor. Thus serum progesterone and serum prolactin levels
should be monitored during treatment and if elevated anti-progesterone and anti-prolactin
agents should be considered.
ADVERSE REACTIONS
Male: Gynecomastia, excessive frequency and duration of penile erections, oligospermia.
Skin and Appendages: Hirsutism, male pattern baldness and acne, gynecomastia.
Fluid/electrolyte Disturbances: Retention of sodium, chloride, water, potassium, calcium, and
inorganic phosphates.
Gastrointestinal: Nausea, cholestatic jaundice, alterations in liver function tests;
hepatocellular neoplasms, peliosis hepatis. hepatic adenomas, nephritis, and cholestatic
hepatitis.
Hematologic: Suppression of clotting factors II, V, VII, & X: bleeding in patients on anticoagulant therapy. Neurological: Increased libido., headache, anxiety, depression, extreme
agitation, irritability, and generalized paresthesia. Trenbolone may cause severe aggressive
behavior.
Other: Serum lipid changes, hypertension, hypercalcaemia, hypertension, oedema, priapism,
and potentiation of sleep apnea.
DRUG INTERACTIONS
Patients on oral anticoagulant therapy require close monitoring especially when androgens
are started or stopped.
Diabetics: androgens may alter the metabolism of oral hypoglycemic agents or may change
insulin sensitivity in patients with diabetes mellitus which may require adjustment of dosage
of insulin and other hypoglycemic drugs.
May alter metabolism of cyclosporine.
Avoid other hepatotoxic medications.
CONTRAINDICATIONS
Patients with known hypersensitivity to any ingredients in this product. Patients with known
or suspected carcinomas of the breast, testis, or prostate. Patients with severe heart liver
disease or kidney disease or with a history of epilepsy. Products containing androgens
should not be used in women as they may cause virilization and fetal harm.
PRECAUTIONS
Women, children and elderly males should not use it. Trenbolone should be used with
extreme caution and with the lowest dosage and duration of treatment necessary for clinical
response pursuant to the advice of a qualified physician. Because androgens may alter
serum cholesterol concentration, caution should be used when administering these drugs to
patients with a history of myocardial infarction or coronary artery disease. Trenbolone may
increase aggressiveness and cause psychiatric changes. At the first sign of psychiatric
changes discontinue use of trenbolone and contact a physician.
Due to its hepatotoxicity, liver functions should be monitored. Discontinue on signs of
jaundicing.
PATIENT MONITORING
Serum Cholesterol, HDL, LDL, TG. Hemoglobin and Hematocrit, Hepatic function
tests-AST/ALT
Prostatic specific antigen- PSA, Testosterone: total, free, and bioavailable.
Dihydrotestosterone & Estradiol.
Progesterone, Prolactin, Blood Pressure.
Coubuu Male patients over 40 should undergo a digital rectal examination and evaluate PSA
prior to androgen use. Periodic evaluations of the prostate should continue while on
androgen therapy, especially in patients with difficulty in urination or with changes in voiding
habits.
DOSAGE AND ADMINISTRATION
Adult male: 100 – 200 mg injected
IM every 5-7 days for a duration limited to 4 weeks under care of a physician.
PRESENTATION
Trenbolone Enanthate 200 mg/ml, 10 ml multiple dose vial.
STORAGE
Store in a cool dry place between 15 – 25°C Protect from light.

Reviews